Pulmonary Tuberculosis: Clinical Presentation, Diagnostic Challenges, Management, and Outcomes – A Case Report
OthersPage Navigation
Abstract
Pulmonary tuberculosis (PTB) is a chronic airborne infectious disease caused by Mycobacterium tuberculosis and remains a leading cause of morbidity and mortality globally. Despite advances in diagnostics and treatment, delayed recognition and poor treatment adherence continue to fuel transmission and disease burden, particularly in resource-limited settings. The disease typically presents with prolonged cough, fever, night sweats, weight loss, and hemoptysis; however, early manifestations are often nonspecific and mimic other respiratory illnesses, resulting in missed or delayed diagnoses.
We report a confirmed case of pulmonary tuberculosis in a young adult male presenting with chronic cough, hemoptysis, fever, and weight loss. This case highlights the clinical presentation, diagnostic challenges, radiological and microbiological confirmation, infection control considerations, management strategies, and clinical outcome. The report underscores the importance of maintaining a high index of suspicion in endemic settings, utilizing rapid molecular diagnostics, ensuring adherence to standardized anti-tubercular therapy, and implementing public health measures to prevent transmission and improve outcomes.
Introduction
Tuberculosis (TB) is an ancient yet persistent infectious disease caused by the acid-fast bacillus Mycobacterium tuberculosis. It primarily affects the lungs but can involve extrapulmonary sites, including lymph nodes, pleura, bones, meninges, and genitourinary tract.
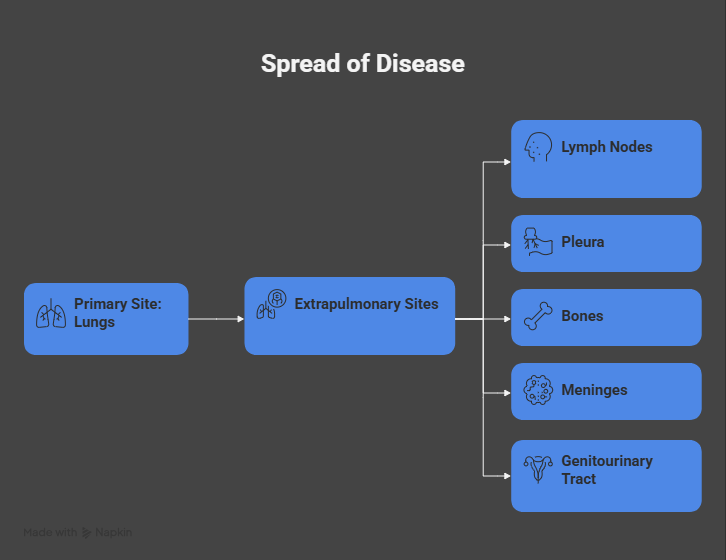
According to global epidemiological estimates, TB remains among the top infectious killers worldwide, disproportionately affecting low- and middle-income countries. Pulmonary tuberculosis is the most common and contagious form of the disease, accounting for the majority of TB-related transmission.
Transmission occurs via inhalation of aerosolized droplet nuclei expelled during coughing, sneezing, speaking, or singing by individuals with active pulmonary disease. The clinical course of PTB varies widely, ranging from asymptomatic or mildly symptomatic disease to severe cavitary lung involvement with respiratory compromise. Host factors such as malnutrition, immunosuppression, human immunodeficiency virus (HIV) infection, diabetes mellitus, and socioeconomic deprivation significantly influence disease susceptibility and progression.
The nonspecific nature of early symptoms frequently leads to misdiagnosis as community-acquired pneumonia or chronic bronchitis, delaying treatment initiation and increasing community transmission. Early microbiological confirmation using sputum-based tests, including smear microscopy and molecular diagnostics such as cartridge-based nucleic acid amplification testing (CBNAAT/GeneXpert), plays a pivotal role in early detection and drug resistance screening. This case report describes a typical presentation of pulmonary tuberculosis, focusing on diagnostic challenges, management principles, and outcome, and highlights the importance of early intervention and adherence to standardized treatment protocols.
Case Report
Patient History
A 34-year-old man presented to the outpatient department with a 6-week history of persistent productive cough associated with yellowish sputum, low-grade intermittent fever, night sweats, and progressive unintentional weight loss of approximately 6 kg. Over the preceding 10 days, he reported intermittent episodes of blood-streaked sputum and one episode of mild hemoptysis. He also complained of generalized fatigue, anorexia, and reduced work capacity.
The patient was a daily-wage construction worker residing in a crowded urban settlement with poor ventilation. He reported close contact with a co-worker who had recently completed treatment for pulmonary tuberculosis. There was no history of previous tuberculosis, diabetes mellitus, chronic lung disease, or immunosuppressive therapy. The patient denied smoking and alcohol consumption. There was no recent travel history.
Clinical Examination
On physical examination, the patient appeared thin and chronically ill. Vital signs revealed a temperature of 38.1°C, pulse rate of 102 beats per minute, blood pressure of 110/70 mmHg, respiratory rate of 22 breaths per minute, and oxygen saturation of 94% on room air.
Respiratory system examination demonstrated reduced air entry with coarse crackles over the right upper lung field and scattered rhonchi.
There was no digital clubbing, cyanosis, or peripheral lymphadenopathy. Cardiovascular and abdominal examinations were unremarkable. There were no signs of pleural effusion.
Based on the chronicity of respiratory symptoms and epidemiological exposure, pulmonary tuberculosis was suspected, and the patient was advised cough hygiene and respiratory isolation precautions while awaiting diagnostic confirmation.
Clinical Evaluation
Differential Diagnosis
The initial differential diagnoses included:
• Pulmonary tuberculosis
• Community-acquired pneumonia
• Bronchogenic carcinoma
• Fungal pulmonary infection
• Bronchiectasis
• Chronic obstructive pulmonary disease with infective exacerbation
The presence of prolonged cough, constitutional symptoms, hemoptysis, and known TB exposure favored pulmonary tuberculosis.
Investigations
Laboratory Studies
Baseline laboratory investigations revealed mild normocytic normochromic anemia (hemoglobin 11.2 g/dL) and elevated erythrocyte sedimentation rate. Total leukocyte count was within normal limits. HIV serology was negative. Liver and renal function tests were normal, allowing initiation of standard anti-tubercular therapy without dose adjustment.
Microbiological and Molecular Testing
Two sputum samples were positive for acid-fast bacilli on Ziehl–Neelsen staining. CBNAAT (GeneXpert MTB/RIF) detected Mycobacterium tuberculosis complex with no evidence of rifampicin resistance, indicating drug-sensitive tuberculosis.

Radiological Evaluation
Chest radiography revealed right upper lobe consolidation with cavitary lesions and fibronodular infiltrates. High-resolution computed tomography of the chest demonstrated thick-walled cavitary lesions with surrounding tree-in-bud nodularity and centrilobular micronodules, consistent with active pulmonary tuberculosis.
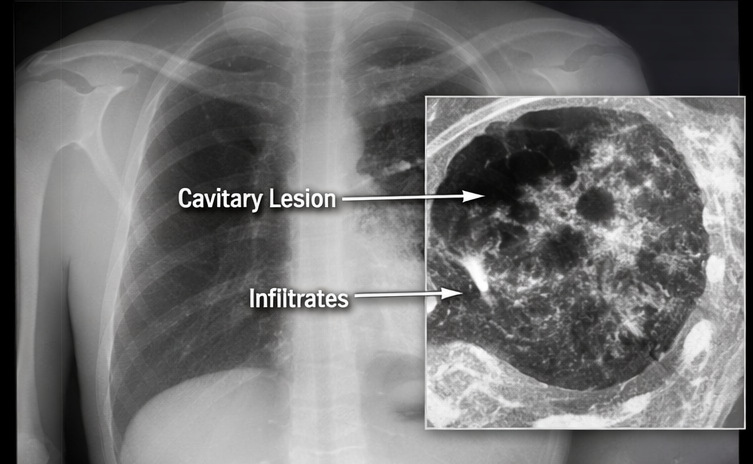
Diagnosis
Based on compatible clinical features, microbiological confirmation of Mycobacterium tuberculosis, and characteristic radiological findings, a definitive diagnosis of drug-sensitive pulmonary tuberculosis was established.
Management and Outcome
Management Strategy
The patient was initiated on standard first-line anti-tubercular therapy under the national tuberculosis control program, comprising isoniazid, rifampicin, pyrazinamide, and ethambutol for the intensive phase (2 months), followed by isoniazid and rifampicin for the continuation phase (4 months), as per weight-based dosing guidelines.
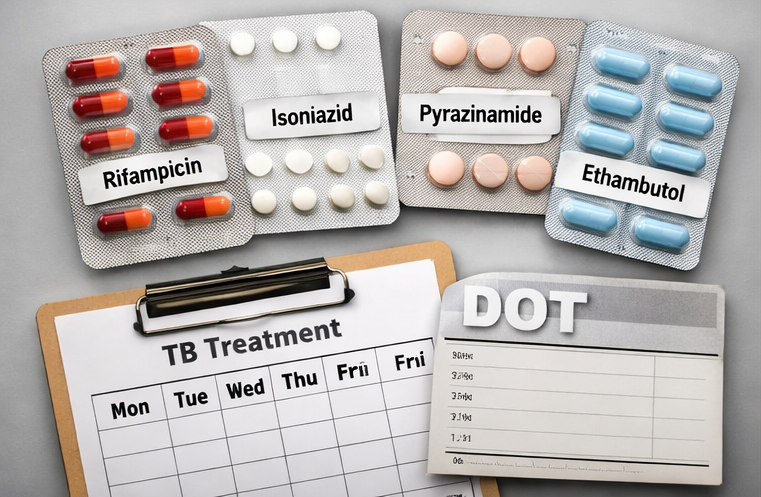
Pyridoxine supplementation was prescribed to prevent isoniazid-induced peripheral neuropathy. The patient received nutritional counseling emphasizing high-protein, calorie-dense meals to address malnutrition. Directly observed treatment (DOT) was implemented to ensure adherence and minimize the risk of treatment default and drug resistance.
Infection control measures, including cough etiquette, mask use during the initial phase of therapy, and household contact screening, were implemented. Close contacts were advised symptom screening and preventive therapy as per public health recommendations.
Follow-Up and Outcome
At the end of the intensive phase (2 months), the patient reported complete resolution of fever and hemoptysis with significant reduction in cough. He had gained 3 kg in body weight. Repeat sputum smear microscopy was negative for acid-fast bacilli, indicating microbiological response.
At completion of the 6-month treatment regimen, the patient was asymptomatic. Follow-up chest radiography demonstrated marked resolution of infiltrates with residual fibrotic scarring. The patient was declared cured based on clinical, radiological, and microbiological criteria.
At a 3-month post-treatment follow-up visit, the patient remained symptom-free with improved exercise tolerance and had returned to full occupational activity.
Discussion
Pulmonary tuberculosis remains a significant clinical and public health challenge, particularly in densely populated regions with socioeconomic vulnerabilities. This case highlights the typical presentation of PTB and emphasizes the importance of epidemiological context in guiding early clinical suspicion. The nonspecific nature of early symptoms frequently leads to misdiagnosis or delayed diagnosis, facilitating ongoing transmission.
Rapid molecular diagnostics such as CBNAAT enable early detection of M. tuberculosis and simultaneous screening for rifampicin resistance, allowing prompt initiation of appropriate therapy. Radiological findings such as upper lobe cavitation and tree-in-bud nodularity are highly suggestive of active disease but are not diagnostic in isolation.
Standardized combination therapy is highly effective when adherence is ensured. Treatment non-adherence is a major driver of multidrug-resistant tuberculosis (MDR-TB), which poses substantial therapeutic and public health challenges. Nutritional support, patient education, and directly observed therapy are critical components of comprehensive TB care.
Post-tuberculosis lung disease, characterized by residual fibrosis, bronchiectasis, and chronic respiratory impairment, is increasingly recognized as a cause of long-term morbidity. Structured follow-up and pulmonary rehabilitation may improve long-term functional outcomes.
Conclusion
Pulmonary tuberculosis is a potentially curable yet highly transmissible disease requiring early clinical suspicion, microbiological confirmation, and strict adherence to standardized multi-drug therapy. This case demonstrates favorable outcomes with timely diagnosis, rapid molecular testing, and supervised treatment. Strengthening diagnostic capacity, ensuring treatment adherence, and implementing public health interventions remain central to tuberculosis control and elimination strategies.
References
- World Health Organization. Global tuberculosis report 2023. Geneva: WHO; 2023.
- Pai M, Behr MA, Dowdy D, Dheda K, Divangahi M, Boehme CC, et al. Tuberculosis. Nat Rev Dis Primers. 2016;2:16076.
- Lawn SD, Zumla AI. Tuberculosis. Lancet. 2011;378(9785):57–72.
- Steingart KR, Schiller I, Horne DJ, Pai M, Boehme CC, Dendukuri N. Xpert MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2014;1:CD009593.
- Nahid P, Dorman SE, Alipanah N, Barry PM, Brozek JL, Cattamanchi A, et al. Official ATS/CDC/IDSA clinical practice guidelines: treatment of drug-susceptible tuberculosis. Clin Infect Dis. 2016;63(7):e147–95.
- Zumla A, Raviglione M, Hafner R, von Reyn CF. Tuberculosis. N Engl J Med. 2013;368(8):745–55.
- Tiberi S, du Plessis N, Walzl G, Vjecha MJ, Rao M, Ntoumi F, et al. Tuberculosis: progress and advances in development of new drugs, treatment regimens, and host-directed therapies. Lancet Infect Dis. 2018;18(7):e183–98.
- Dheda K, Barry CE, Maartens G. Tuberculosis. Lancet. 2016;387(10024):1211–26.
- Migliori GB, Tiberi S, Zumla A, Petersen E, Chakaya JM, Wejse C, et al. MDR/XDR-TB management of patients and contacts. Eur Respir J. 2020;56(3):2001639.
- Allwood BW, van der Zalm MM, Amaral AFS, Byrne A, Datta S, Egere U, et al. Post-tuberculosis lung disease: clinical review. Lancet Respir Med. 2021;9(5):536–54.
Read more such content on @ Hidoc Dr | Medical Learning App for Doctors
Recommended News For You
Recommended Articles For You
Featured News
Featured Articles
Featured Events
Featured KOL Videos
1.
Remote monitoring can improve recovery from cancer surgery
2.
Intractable cancers may respond better to treatment when using new radiation and high-performance computing.
3.
A Win for AI in Cancer; 2025's Transformative Drugs; FDA Clarifies 'Underway' Trials
4.
Conditional EU Nod for Weekly Pill in Pediatric Glioma
5.
high response rate when using a bispecific antibody to treat R/R multiple myeloma.
1.
The Technological Revolution in Precision Oncology and Tumor Microenvironment Therapy
2.
The Role of the Oncology Pharmacist: From Treatment to Trials and Beyond
3.
Unlocking the Secrets of Neutrophils: Exploring Their Role in Immune Defense
4.
New Hope for Rectal Cancer Patients: Breakthrough Drug Shows Promising Results
5.
Unveiling the Mystery of Echinocyte: A Closer Look at the Unique Red Blood Cell
1.
International Lung Cancer Congress®
2.
Genito-Urinary Oncology Summit 2026
3.
Future NRG Oncology Meeting
4.
ISMB 2026 (Intelligent Systems for Molecular Biology)
5.
Annual International Congress on the Future of Breast Cancer East
1.
Role of Nimotuzumab in Management of Nasopharyngeal Cancer
2.
The Landscape of First-Line Treatment for Urothelial Carcinoma- The Conclusion
3.
Pazopanib Takes Center Stage in Managing Renal Cell Carcinoma - Part III
4.
A Comprehensive Guide to First Line Management of ALK Positive Lung Cancer - Part IV
5.
An Eagles View - Evidence-based Discussion on Iron Deficiency Anemia- Panel Discussion
© Copyright 2026 Hidoc Dr. Inc.
Terms & Conditions - LLP | Inc. | Privacy Policy - LLP | Inc. | Account Deactivation

